 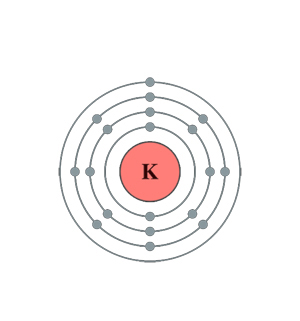
Negative ions are anions.Ĭomplete the following table and consider how you would explain your answers if you were teaching this concept to a peer. When there are more electrons than protons, as in the case of the sulfur ion, the net charge is negative. If, however, there are fewer electrons than protons as in the case of the potassium ion, the net charge is positive since there is one more positive proton than negative electrons. potassium ( K ) floated to the top to form Earth's crust, leaving the Fe. Therefore, the M shell of the Potassium atom only. on the left, 6 protons and 6 neutrons in the nucleus with 6 electrons in. The M shell of atoms having an atomic number below 20 can hold only 8 electrons in their M shell. Name of Nuclear Atomic Mass Element Symbol Number Number Protons Neutrons Electrons Scandium 4521Sc 21 45 21 24 21 Germanium 7632Ge 32 76 32 44 32 Tin. K, L, M, and N shells as 2, 8, 8, and 1, respectively. The electrons are housed in four shells i.e. Since an electron has the same magnitude of charge as a proton but opposite sign (electrons are negative), in order for an atom to be neutral, to have no net charge, the number of protons and electrons are equal to each other. The final Bohr model of the Potassium atom consists of 19 protons, 20 neutrons, and 19 electrons. Protons are positively charged particles and the number of protons is determined by the atomic number of the element. If the number of electrons changes compared to the number in a neutral atom, the resulting particle is charged. To find the number of neutrons, subtract the number of protons from the mass number. The mass number, 65, is the sum of the protons and the neutrons. However, the number of electrons in an atom can change. For all atoms with no charge, the number of electrons is equal to the number of protons. Since the iodine is added as a 1 anion, the number of electrons is 54. Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 53 74). The nucleus of an atom of potassium contains 19 protons and 20 neutrons. In a neutral atom, the number of protons and electrons are equal to each other. The atomic number of iodine (53) tells us that a neutral iodine atom contains 53 protons in its nucleus and 53 electrons outside its nucleus. 
What is the mass number of this isotope 18 Different of an element have different numbers of neutrons. How many neutrons make up one of these potassium atoms 20 One isotope of sodium has 11 protons and 7 neutrons. One of the most stable types of potassium atoms has the mass number 39. If the number of protons changes (as you will in see in unit 2 can happen during nuclear reactions) the identity of the atom changes. All atoms of the element potassium have 19 protons. \)Īs discussed in the previous section, the number of protons in an atom determines to which element an atom belongs. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
